Introduction
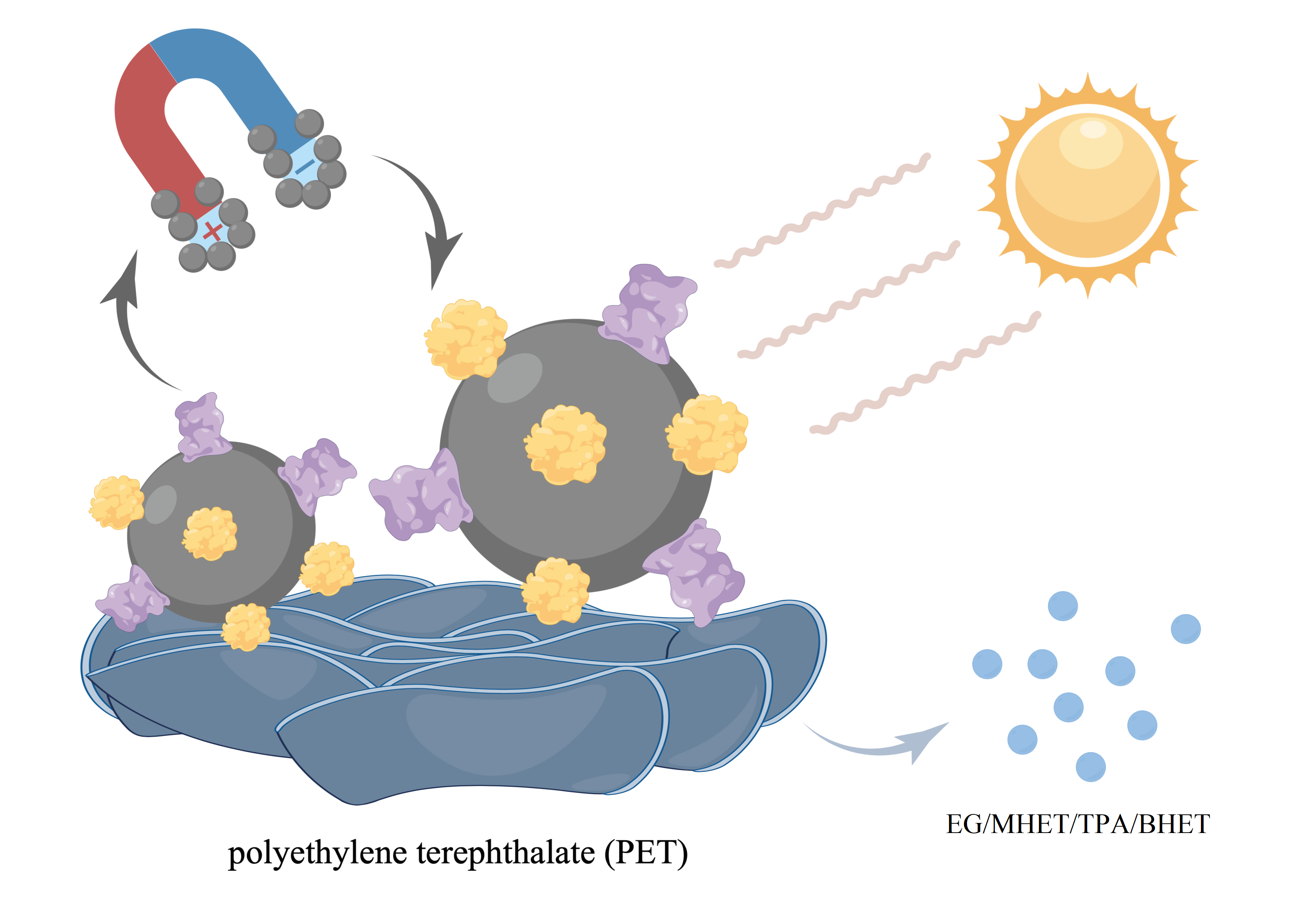
Scheme. PET degradation and Co-immobilized enzyme 4M-MG8@Fe3O4NPs recycle.
Recent years, the enormous waste plastic has become a severe environmental issue with the wide abuse of plastic products. Waste plastic showed a great negative impact on the ecological environment[1] because its degradation time is more than 100 years, even 500 years[2]. In addition, plastic waste may put a threat to human health[3][4]. For instance, it has been reported that microplastics smaller than 5mm can accumulate in the human body in different ways. Polyethylene terephthalate (PET) accounts for 12% of global solid waste[2]. The traditional methods of PET waste disposal are incineration and landfills[5]. However, a large number of toxic gases such as dioxins[6], and carbon monoxide produced by incineration will cause damage to the atmosphere. Meanwhile, landfill treatment will occupy land resources and cause severe contamination of groundwater[7].
Fortunately, biodegradation of PET plastic has been discovered. Therefore, circular carbon economy for PET could theoretically be achieved through rapid enzymatic depolymerization followed by repolymerization or conversion into other products[8]. Enzymatic degradation of PET has become a hit for its environmental friendliness and sustainability[9].
It has been reported various PET enzymes have excellent properties for the biodegradation of PET plastics[10].In 2016, Yoshida et al. derived PET hydrolase (PETase) from Ideonella sakaiensis[11], which can effectively degrade PET into bis (2-hydroxyethyl) terephthalic (BHET), mono (2-hydroxyethyl) terephthalate (MHET), TPA, and EG. At moderate temperature (30–40◦C), due to its unique PET substrate binding pocket[12]. IsPETase has immediately become one of the most promising PET plastic degradation enzymes. However, this poor thermal stability of wild-type IsPETase limited its application for PET degradation[13]. Until now, the improvement of thermal stability and biodegradation capacity is the main research direction of PETase. And many kinds of engineering enzyme are based on IsPETase[14][15].
We proposed solar-driven enzymatic PET degradation with DuraPETase-4M[16], a high-efficiency mutant of PETase, and MG8[17], a new PET hydrolase from the human saliva metagenome, by using Fe3O4 nanoparticles (NPs) as a solar-to-thermal convertor. Solar irradiation could elevate the temperature of Fe3O4 NPs for the effective PET degradation[18][19]. Also, by covalently binding to Fe3O4 NPs, enzymes can easily be recycled, which will make a contribution to cost reduction. DuraPETase-4M is mainly good at the degradation of amorphous PET[16],while MG8 shows excellent catalytic activity of crystalline PET[17].By combining them, PET can be more thoroughly degraded.
References
[1] Papadopoulou A, Hecht K, Buller R. Enzymatic PET degradation. Chimia (Aarau), 2019, 73: 743–749.
[2] D. Zhou, J. Chen, J. Wu, et al. Biodegradation and catalytic-chemical degradation strategies to mitigatemicroplastic pollution, Sustain. Mater. Technol, 2021, 28.
[3] Lu H, Diaz DJ, Czarnecki NJ, et al. Machine learning-aided engineering of hydrolases for PET depolymerization. Nature, 2022, 604(7907): 662-667.
[4] H.F. Son, I.J. Cho, S. Joo, et al. Rational protein engineering of thermo-stable PETase from Ideonella sakaiensis for highly efficient PET degradation. ACS Catal, 2019, 9: 3519–3526.
[5] Demetrious A, Crossin E. Life cycle assessment of paper and plastic packaging waste in landfill, incineration, and gasification-pyrolysis. Mater. Cycles Waste Manag, 2021, 850–860.
[6] Chen X, Yan N. A brief overview of renewable plastics. Mater Today Sustain, 2020, 7–8: 100031.
[7] Chu. J, Cai. Y, Li. C, et al. Dynamic flflows of polyethylene terephthalate (PET) plastic in China. Waste Manag, 2021, 124: 273–282.
[8] R. Koshti, L. Mehta, N. Samarth. Biological recycling of polyethylene terephthalate: a mini-review. Polym. Environ, 2018, 26: 3520–3529.
[9] Amobonye A, Bhagwat P, Singh S, et al. Plastic biodegradation: frontline microbes and their enzymes. Sci Total Environ, 2021, 759: 143536.
[10] Then J, Wei R, Oeser T, et al. Ca2+ and Mg2+ binding site engineering increases the degradation of polyethylene terephthalate fifilms by polyester hydrolases from Thermobififida fusca. Biotechnol, 2015, 10:592–598.
[11] Shosuke Yoshida, KH, Takehana, et al. A bacterium that degrades and assimilates poly (ethylene terephthalate). Science, 2015, 351: 1196–1199.
[12] Han X, Liu W, Huang, JW, et al. Structural insight into catalytic mechanism of PET hydrolase. Nat Commun, 2017, 8: 2106.
[13] Son HF, Cho IJ, Joo S, et al. Rational protein engineering of thermo-stable PETase from Ideonella sakaiensis for highly effificient PET degradation. ACS Catal, 2019, 9: 3519–3526.
[14] Yinglu Cui, Yanchun Chen, Xinyue Liu, et al. Computational Redesign of a PETase for Plastic Biodegradation under Ambient Condition by the GRAPE Strategy. ACS Catal, 2021, 11: 1340−1350
[15] Fusako Kawai, Takeshi Kawabata, Masayuki Oda. Current State and Perspectives Related to the Polyethylene Terephthalate Hydrolases Available for Biorecycling. ACS Sustainable Chem Eng, 2020, 8: 8894−8908.
[16] Liu Y, Liu Z, Guo Z, et al. Enhancement of the degradation capacity of IsPETase for PET plastic degradation by protein engineering. Sci Total Environ, 2022, 834: 154947.
[17] Eiamthong B, Meesawat P, Wongsatit T, et al. Discovery and Genetic Code Expansion of a Polyethylene Terephthalate (PET) Hydrolase from the Human Saliva Metagenome for the Degradation and Bio-Functionalization of PET. Angew Chem Int Ed Engl, 2022, 3: e202203061.
[18] M. Chu, Y. Shao, J. Peng, et al. Near-infrared laser light mediated cancer therapy by photothermal effect of Fe3O4 magnetic nanoparticles. Biomaterials, 2013, 34: 4078–4088.
[19] Li Y, Chen L, Yao, et al. Fe3O4/PVDF-HFP photothermal membrane with in-situ heating for sustainable, stable and efficient pilot-scale solar driven membrane distillation. Desalination, 2020, 478.




